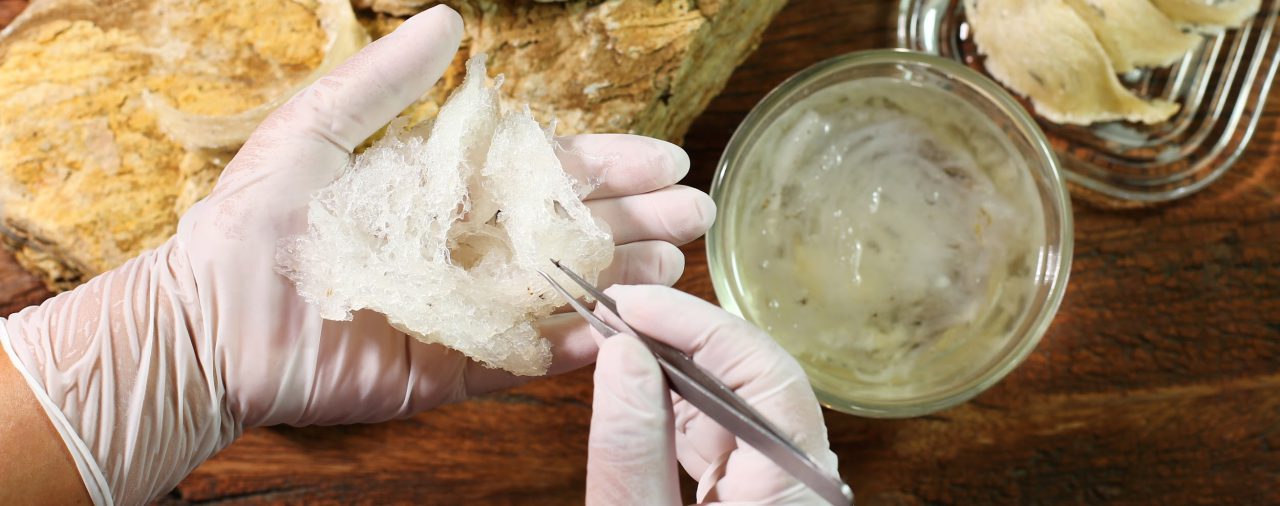
Known as the ‘Caviar of the East’, edible bird’s nest has been part of Chinese traditional medicine for more than 100 years and is said to carry a lot of nutritional and medicinal properties. It is made almost entirely from the gelatinous salivary secretion containing highly concentrated mucin glycoproteins by swiftlets native to the Indo-Pacific region.
There are more than 24 species of insectivorous, echolocating swiftlets distributed around the world, but only a few produce nests that are deemed ‘edible’. The nests are built almost exclusively by the male swiftlets over a period of approximately 35 days and weigh 1–2 times their actual body weight.1
Due to its limited supply and enormous demand, edible bird’s nest is reputably expensive. Consequently, many fake or adulterated products are available in the market. It is often difficult to detect adulterated edible bird’s nest by simple physical means especially when the adulteration level is no more than 10% by weight.
Among the adulterants used are coralline seaweed, tremella fungus, agar, fish bladder and pork rind which can be detected by microscope examination. Other more subtle forms of adulterants are water soluble and can be absorbed by the bird’s nest such as glucose, sucrose, hydrolyzed marine collagen and monosodium glutamate (MSG). These types of adulterants have the same appearance as the unadulterated nest under the microscope.
Various physical, analytical and chemical techniques have been designed to check for the presence of adulterants in the edible bird’s nest. Unfortunately, most of the techniques are invasive, require tedious sample preparation, slow and generally not easy to use. More simple and non-invasive ways that are reported include:
Fourier-Transform Infrared (FTIR) technology
The Fourier-transform infrared (FTIR) technology utilizes infrared radiation (IR) to show the differing “fingerprints” of samples being analysed and hence provides insight into their chemical structures. Using this, scientist can identify if the sample is made of organic, inorganic or polymeric material and more specifically what the sample is by comparing with existing reference spectra.
Every compound has its own unique light absorption ability which is captured, measured and represented by the equipment as a series of different peak patterns. Genuine bird’s nest also has its own unique peak pattern. Comparing the sample peak pattern (from 50 times measurement) with the reference standard genuine bird’s nest peak pattern allows us to differentiate the sample as either genuine or fake bird’s nest. In the industry, FTIR values above 0.9500 are deemed genuine bird’s nest and the standard reference FTIR value is 1.0000.
Raman Microspectroscopy Technique
Among the techniques, vibrational spectroscopic methods can provide a simple, non-invasive and fast way to detect the adulterants. Spectroscopy deals with the interaction between photons of radiation and molecules. Measurement of the response pattern can give information about molecular structure; on what and how much is present in a food product.2
A previously used method, infrared spectroscopy was able to detect large adulterants but may not be sensitive enough to detect subtle adulterants. Raman spectroscopy is a particularly useful technique that is complementary to IR spectroscopy. It involves no sample preparation and most importantly allows measurements in the presence of moisture.3 Raman spectroscopy is often able to detect adulterants better than the more commonly used IR spectroscopy. It is non-invasive, applicable to opaque samples, is not affected by moisture in the sample and is used as an authentication method for edible bird’s nest.4 However, this technology tends to be a bit more costly than IR spectroscopy so IR spectroscopy is generally used commercially.
Thermal analysis method
Thermal analysis consists of a group of techniques in which a physical property of a substance is measured in a controlled temperature program. The unique thermal properties of edible bird’s nest serve as analytical standards and offer a simple, fast technique that requires only a small sample size to authenticate, and check for edible adulterants which may be introduced into it. 7
Thermogravimetry, differential thermogravimetry, and differential scanning calorimetry are other methods used for the rapid identification and authentication of edible bird’s nest. Thermogravimetry is a technique that measures the increase or decrease in the sample’s weight when heated, while scanning calorimetry measures the amount of heat required to increase the temperature of a sample.5,6
The presence of adulterants in the bird’s nest will be detected by different sets of curves produce from their thermal analysis reading and compared to curve produced by the genuine bird’s nest. The use of both Raman microspectroscopy and thermal analysis are highly suggestive steps to fight food fraud with science.
References:
1 Marcone M. Characterization of the edible bird’s nest the “Caviar of the East”. Food Research International. 2005;38(10):1125-1134. doi:10.1016/j.foodres.2005.02.008
2 Downey G. Food Authentication Using Vibrational Spectroscopic Techniques. Published 2015. Accessed December 19, 2018.
3 ‘Raman Spectroscopy – an Overview | ScienceDirect Topics’, accessed 5 December 2018.
4 Shim E, Chandra G, Pedireddy S, Lee S. Characterization of swiftlet edible bird nest, a mucin glycoprotein, and its adulterants by Raman microspectroscopy. J Food Sci Technol. 2016;53(9):3602-3608. doi:10.1007/s13197-016-2344-3
5 Kodre K, Attarde S, Yendhe P, Patil R, Barge V. Differential Scanning Calorimetry: A Review. J Pharm Anal. 2014;3(3):11-22. Accessed December 19, 2018.
6 A Beginners Guide Thermogravimetric Analysis (TGA). Perkinelmer.com. Published 2010. Accessed December 19, 2018.
7 Shim E, Chandra G, Lee S. Thermal analysis methods for the rapid identification and authentication of swiftlet ( Aerodramus fuciphagus ) edible bird’s nest – A mucin glycoprotein. Food Research International. 2017;95:9-18. doi:10.1016/j.foodres.2017.02.018